A prediction model for enteral nutrition interruption after gastric cancer surgery established using machine learning algorithms
-
摘要:
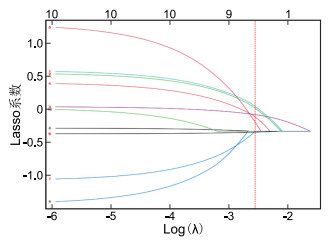
目的 胃癌患者在术后肠内营养中断(ENI)风险较高,但关于此类患者术后ENI的影响因素尚无统一意见,本研究旨在基于临床资料,利用机器学习算法构建能个体化预测胃癌患者术后ENI风险的模型,为ENI的评估及防治提供指导。 方法 选取2023年5月—2025年4月南京医科大学第一附属医院普外胃外科一、二病区收治的胃癌患者190例,按7∶3随机分为训练集133例和验证集57例。190例胃癌患者均行胃癌根治术治疗。采用逻辑回归(LR)、随机森林(RF)、支持向量机(SVM)3种机器学习算法构建预测模型,并分析模型性能。采用验证集数据验证最优模型效能。 结果 LASSO回归筛选出的胃癌术后ENI相关变量包括年龄、术后24 h VAS评分、术后首次下床时间、术后第1天下床活动距离、术后并发症情况、肿瘤分期、抗生素应用种类。LR、RF、SVM三种模型的AUC值分别为0.768、0.893、0.861,RF模型的预测效能更优。RF模型中风险阈值范围为0.03~0.95,在此范围内对依据RF模型预测结果实施相应措施能为ENI预防计划提供显著的净效益。基于验证集数据及LASSO回归筛选出的胃癌术后ENI相关变量构建RF模型的AUC值为0.853。 结论 基于机器学习算法建立的3种模型预测表现均较好,尤其是RF模型预测效能更优,可有效判断胃癌术后ENI发生风险,识别高风险患者。 Abstract:Objective Gastric cancer patients face a high risk of enteral nutrition interruption (ENI) after surgery. However, no consensus has been reached regarding the factors influencing postoperative ENI. This study aims to develop an individualized risk prediction model for postoperative ENI in gastric cancer patients using machine learning algorithms based on clinical data, thereby providing guidance for the assessment, prevention, and management of ENI. Methods A total of 190 patients with gastric cancer were recruited from Ward 1 and Ward 2 of the Department of General Surgery (Gastric Surgery Division) at the First Affiliated Hospital of Nanjing Medical University between May 2023 and April 2025. Patients were randomly divided into a training set (n=133) and a validation set (n=57) in a ratio of 7∶3. Three machine learning algorithms, namely logistic regression (LR), random forest (RF), and support vector machine (SVM), were adopted to construct prediction models, and model performance were analyzed. The optimal model performance was verified using the validation set data. Results LASSO regression identified the following variables associated with ENI after gastric cancer surgery: age, VAS score at 24 hours after surgery, time to first postoperative ambulation, distance of ambulation on postoperative day 1, postoperative complications, tumor stage, and types of antibiotics. The AUC values of the LR, RF, and SVM models were 0.768, 0.893, and 0.861, respectively, with the RF model demonstrating superior predictive performance. The risk threshold range of the RF model was 0.03 to 0.95, within which implementing targeted interventions based on the prediction results of the RF model can provide substantial net clinical benefits for ENI prevention. Using the validation set, the RF model constructed with LASSO-selected variables achieved an AUC of 0.853. Conclusion The prediction performances of the three models established based on machine learning algorithms were all satisfactory. In particular, the RF model demonstrated superior predictive efficiency, enabling effective assessment of ENI risk after gastric cancer surgery and reliable identification of high-risk patients. -
表 1 190例胃癌术后ENI发生情况
Table 1. Incidence of ENI in 190 cases after gastric cancer surgery
ENI原因 例数 占比(%) 喂养不耐受 69 74.19 外出检查 10 10.75 患者依从性差 4 4.30 管道因素(堵管、拔管等) 4 4.30 医护操作 6 6.45 合计 93 100.00 表 2 2组胃癌患者基线资料比较
Table 2. Comparison of characteristics between the two groups of gastric cancer patients
项目 ENI组(n=93) Non-ENI组(n=97) 统计量 P值 性别[例(%)] 0.003a 0.958 男性 55(59.14) 57(58.76) 女性 38(40.86) 40(41.24) 年龄(x±s,岁) 63.62±5.89 61.29±6.02 2.695b 0.008 合并基础疾病[例(%)] 高血压 29(31.18) 28(28.87) 0.121a 0.728 糖尿病 20(21.51) 8(8.25) 6.642a 0.010 冠心病 12(12.90) 11(11.34) 0.109a 0.741 ASA分级[例(%)] 0.509a 0.611 Ⅰ级 18(19.35) 20(20.62) Ⅱ级 52(55.91) 57(58.76) Ⅲ级 23(24.73) 20(20.62) 肿瘤分期[例(%)] 3.126c 0.002 Ⅰ期 22(23.66) 43(44.33) Ⅱ期 46(49.46) 42(43.30) Ⅲ期 25(26.88) 12(12.37) 淋巴结转移[例(%)] 0.269a 0.604 有 76(81.72) 82(84.54) 无 17(18.28) 15(15.46) 根治方式[例(%)] 0. 201a 0.904 全胃切除术 31(33.33) 35(36.08) 近端胃切除术 15(16.13) 16(16.49) 远端胃切除术 47(50.54) 46(47.42) 手术方法[例(%)] 5.049a 0.025 开腹 9(9.68) 2(2.06) 腹腔镜 84(90.32) 95(97.94) 手术时间(x±s,min) 203.98±29.16 198.02±30.40 1.378b 0.170 术中出血量(x±s,mL) 147.66±42.82 153.82±39.88 1.027b 0.306 术后24 h VAS评分(x±s,分) 3.62±0.81 3.24±0.77 3.294b 0.001 术后首次下床时间(x±s,h) 22.87±2.53 20.50±2.98 5.898b <0.001 术后第1天下床活动距离(x±s,m) 104.30±13.54 111.72±14.01 3.710b <0.001 术后并发症情况[例(%)] 9.309c 0.010 0种 60(64.52) 80(82.47) 1种 24(25.81) 15(15.46) 2种及以上 9(9.68) 2(2.06) 术前行新辅助化疗[例(%)] 1.449a 0.229 有 24(25.81) 18(18.56) 无 69(74.19) 79(81.44) 抗生素应用种类[例(%)] 7.545c 0.023 0种 58(62.37) 77(79.38) 1种 28(30.11) 18(18.56) 2种及以上 7(7.53) 2(2.06) 肠内营养开始时间[例(%)] 6.268a 0.012 术后24 h 40(43.01) 25(25.77) 术后48 h 53(56.99) 72(74.23) 注:a为χ2值,b为t值,c为U值。 表 3 训练集中3种模型对胃癌术后ENI的预测效能
Table 3. Predictive performance of three models for ENI after gastric cancer surgery in the training set
模型 AUC 灵敏度(%) 特异度(%) 准确度(%) 精确度(%) F1值 LR 0.768 91.40 65.98 78.42 72.03 0.844 RF 0.893 84.95 87.63 86.32 86.81 0.856 SVM 0.861 76.34 87.63 82.10 85.54 0.791 -
[1] 中国抗癌协会肿瘤营养专业委员会, 中华医学会肠外肠内营养学分会. 胃癌患者的营养治疗专家共识[J]. 肿瘤代谢与营养电子杂志, 2023, 10(2): 208-212.Oncology Nutrition Professional Committee of the Chinese Anti-Cancer Association, Parenteral and Enteral Nutrition Society of the Chinese Medical Association. Expert consensus on nutritional treatment for patients with gastric cancer[J]. Journal of Tumor Metabolism and Nutrition(Electronic Edition), 2023, 10(2): 208-212. [2] ZHENG H Y, CAI L N, WANG P R, et al. Causes and impacts of interrupted enteral nutrition in critically ill patients: a secondary analysis of a cluster-randomized controlled trial[J]. Nurs Crit Care, 2025, 30(2): e70006. DOI: 10.1111/nicc.70006. [3] VAN NIEUWKOOP M M, RAMNARAIN D, POUWELS S. Enteral nutrition interruptions in the intensive care unit: a prospective study[J]. Nutrition, 2022, 96: 111580. DOI: 10.1016/j.nut.2021.111580. [4] 余善招, 包益萍, 赵灵, 等. 基于机器学习算法的成人神经外科重症患者肠内营养喂养不耐受风险预测模型的构建[J]. 中华全科医学, 2025, 23(3): 414-416, 452. doi: 10.16766/j.cnki.issn.1674-4152.003916YU S Z, BAO Y P, ZHAO L, et al. Construction of risk prediction model of enteral feeding intolerance in severe neurosurgical patients based on machine learning algorithm[J]. Chinese Journal of General Practice, 2025, 23(3): 414-416, 452. doi: 10.16766/j.cnki.issn.1674-4152.003916 [5] 中华医学会肿瘤学分会, 中华医学会杂志社. 中华医学会胃癌临床诊疗指南(2021版)[J]. 中华医学杂志, 2022, 102(16): 1169-1189.Chinese Society of Oncology, Chinese Medical Association; Chinese Medical Association Publishing House. Chinese society of oncology clinical practice guidelines for gastric cancer (2021 edition)[J]. Chinese Medical Journal, 2022, 102(16): 1169-1189. [6] 中华医学会外科学分会腹腔镜与内镜外科学组, 中国医师协会外科医师分会上消化道外科医师专家工作组, 《中华消化外科杂志》编辑委员会. 精准胃癌外科诊疗中国专家共识(2024版)[J]. 中华消化外科杂志, 2024, 23(3): 323-333.Laparoscopic and Endoscopic Surgery Group, Chinese Society of Surgery, Chinese Medical Association, Expert Working Group on Upper Gastrointestinal Surgery, Chinese College of Surgeons, Chinese Medical Doctor Association, Editorial Board of Chinese Journal of Digestive Surgery. Chinese expert consensus on precision surgical diagnosis and treatment of gastric cancer (2024 edition)[J]. Chinese Journal of Digestive Surgery, 2024, 23(3): 323-333. [7] 中华医学会肠外肠内营养学分会. 中国成人患者肠外肠内营养临床应用指南(2023版)[J]. 中华医学杂志, 2023, 103(13): 946-974.Parenteral and Enteral Nutrition Society, Chinese Medical Association. Guideline for clinical application of parenteral and enteral nutrition in adults patients in China (2023 edition)[J]. Chinese Medical Journal, 2023, 103(13): 946-974. [8] MUSCARITOLI M, ARENDS J, BACHMANN P, et al. ESPEN practical guideline: Clinical Nutrition in cancer[J]. Clin Nutr, 2021, 40(5): 2898-2913. doi: 10.1016/j.clnu.2021.02.005 [9] LI Y, ZENG X, ZHOU L. Ultrasound-guided peripheral nerve radiofrequency ablation for craniofacial postherpetic neuralgia: efficacy and safety in a retrospective cohort[J]. J Clin Neurosci, 2025, 138: 111408. DOI: 10.1016/j.jocn.2025.111408. [10] XU Y Y, HU Q Y, PEI D D, et al. Construction of a preoperative emotional state and postoperative intra-abdominal pressure based prediction model for early enteral feeding intolerance in postoperative patients with gastric cancer[J]. Front Nutr, 2024, 11: 1480390. DOI: 10.3389/fnut.2024.1480390. [11] LIU Z K, MA W X, ZHANG J J, et al. Risk factor analysis and establishment of a predictive model for complications of elderly advanced gastric cancer with Clavien-Dindo classification≥Ⅱ grade[J]. BMC Cancer, 2024, 24(1): 1185-1198. doi: 10.1186/s12885-024-12965-5 [12] 徐燕飞, 王寿华, 施伟斌, 等. 口服肠内营养粉剂及综合干预促进胃癌术后康复的临床疗效[J]. 新医学, 2022, 53(11): 838-841.XU Y F, WANG S H, SHI W B, et al. The clinical efficacy of oral enteral nutrition powder and comprehensive intervention in promoting postoperative recovery after gastric cancer surgery[J]. Journal of New Medicine, 2022, 53(11): 838-841. [13] 李粉红, 卫亚楠, 吴梦丽, 等. 加速康复外科理念在老年头颈癌病人围术期营养管理中的应用实践[J]. 实用老年医学, 2025, 39(2): 134-138.LI F H, WEI Y N, WU M L, et al. Enhanced recovery after surgery (ERAS) principles in nutritional management for elderly patients with head and neck cancer perioperatively[J]. Practical Geriatrics, 2025, 39(2): 134-138. [14] WANG J, WANG L, ZHAO M, et al. Effect of early enteral nutrition support combined with chemotherapy on related complications and immune function of patients after radical gastrectomy[J]. J Healthc Eng, 2022: 1531738. DOI: 10.1155/2022/1531738. [15] HUA X, LIU Y, ZHOU Y. Clinical effect of tumor-specific total nutrients in patients with adjuvant chemotherapy after radical gastric cancer resection: a randomized controlled trial[J]. J Gastrointest Cancer, 2024, 55(2): 845-851. doi: 10.1007/s12029-024-01029-3 [16] CHERNEVSKAYA E, BELOBORODOVA N, KLIMENKO N, et al. Serum and fecal profiles of aromatic microbial metabolites reflect gut microbiota disruption in critically ill patients: a prospective observational pilot study[J]. Crit Care, 2020, 24(1): 312-324. doi: 10.1186/s13054-020-03031-0 [17] 李海菊, 李龙倜, 王娜, 等. 重型颅脑损伤患者喂养不耐受的影响因素分析[J]. 重庆医学, 2025, 54(1): 172-175, 180.LI H J, LI L T, WANG N, et al. Analysis on influencing factors of feeding intolerance in patients with severe traumatic brain injury[J]. Chongqing Medicine, 2025, 54(1): 172-175, 180. -





 下载:
下载: