The relationship of RAB24 expression in gastric cancer with clinical prognosis and immune infiltration
-
摘要:
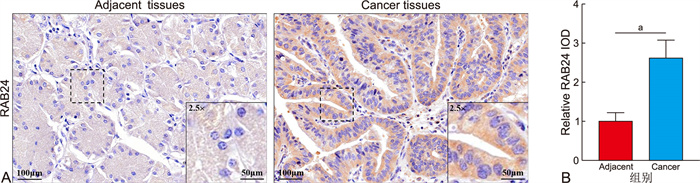
目的 探讨RAB24在胃癌患者组织中的表达情况及与临床预后的关系,并深入分析其调控癌细胞免疫浸润的作用。 方法 纳入2013年2月—2018年3月期间于蚌埠医科大学第一附属医院接受胃癌根治术的105例患者,采用免疫组织化学染色方法分析胃癌中RAB24的表达情况并探讨其与患者生存期之间的关系。采用ROC曲线、单因素及多因素分析研究RAB24对患者术后5年生存率的预测价值及影响患者生存情况的危险因素。GO及GSEA预测RAB24在胃癌中的生物学功能。网络生物信息学分析RAB24表达与免疫浸润和免疫细胞的关系。 结果 RAB24在胃癌组织中高表达,且其表达与患者预后不良相关(P<0.05),并具有良好的预后预测价值(AUC=0.844,P<0.05)。Cox回归分析结果显示,RAB24高表达、CEA≥5 μg/L、CA199≥37 kU/L、T3~4期、N2~3期是影响胃癌患者术后5年生存率的独立危险因素(P<0.05)。基因富集分析提示,RAB24可能参与免疫细胞浸润等生物学过程。免疫浸润相关性分析结果显示,RAB24表达水平与免疫浸润呈正相关关系(P<0.05);免疫细胞相关性分析结果显示,RAB24表达水平与巨噬细胞、髓源性抑制细胞及17型辅助T细胞呈正相关关系(P<0.05),与效应记忆CD4+ T细胞、2型辅助T细胞及记忆B细胞呈负相关关系(P<0.05)。 结论 RAB24在胃癌组织中高表达,其可能通过作用于免疫细胞浸润,进而影响胃癌患者的远期预后。 Abstract:Objective To investigate the expression of RAB24 in the tissues of gastric cancer patients and its relationship with clinical prognosis, and to deeply analyze the role of RAB24 in regulating the immune infiltration of cancer cells. Methods A total of 105 patients who underwent radical gastric cancer surgery in First Affiliated Hospital of Bengbu Medical University from February 2013 and March 2018 was included. Immunohistochemical staining methods were used to analyze the expression of RAB24 in gastric cancer and to explore the relationship between it and patient survival. ROC curve, univariate, and multivariate analyses were performed to assess the predictive value of RAB24 for the 5-year postoperative survival rate and to identify risk factors influencing patient survival. GO and GSEA analyses were performed to predict the biological functions of RAB24 in gastric cancer. Bioinformatics online platforms were used to analyze the relationship between levels of RAB24 and immune infiltration as well as immune cells. Results High expression of RAB24 in gastric cancer tissue was significantly associated with poor patient prognosis (P < 0.05) and had a good predictive value for prognosis (AUC=0.844, P < 0.05). The results of Cox regression analysis showed that high expression of RAB24, CEA≥5 μg/L, CA199≥37 kU/L, T3-4 stage, and N2-3 stage were independent risk factors affecting the 5-year survival rate of gastric cancer patients (P < 0.05). Gene enrichment analysis suggested that RAB24 may participate in immune infiltration processes. The results of immune infiltration correlation analysis showed that the expression level of RAB24 was positively correlated with immune infiltration (P < 0.05), while the results of immune cell correlation analysis showed that the expression level of RAB24 was positively correlated with macrophages, myeloid derived suppressor cells, and Th17 cells (P < 0.05), and negatively correlated with effector memory CD4+T cells, Th2 cells, and memory B cells (P < 0.05). Conclusion The high expression of RAB24 in gastric cancer may affect the long-term prognosis of gastric cancer patients by influencing the infiltration of immune cells. -
Key words:
- Gastric cancer /
- RAB24 /
- Bioinformatics /
- Prognosis /
- Immune infiltration
-
表 1 RAB24在胃癌中的水平与临床病理参数间的关系[例(%)]
Table 1. The relationship between the level of RAB24 in gastric cancer and clinical pathological parameters
项目 例数 低表达(n=52) 高表达(n=53) χ2值 P值 性别 男性 74 37(50.0) 37(50.0) 0.023 0.880 女性 31 15(48.4) 16(51.6) 年龄(岁) <60 41 20(48.8) 21(51.2) 0.015 0.903 ≥60 64 32(50.0) 32(50.0) CEA(μg/L) <5 49 31(63.3) 18(36.7) 6.940 0.008 ≥5 56 21(37.5) 35(62.5) CA199(kU/L) <37 47 34(72.3) 13(27.7) 17.720 < 0.001 ≥37 58 18(31.0) 40(69.0) 病理组织学类型 腺癌 66 30(45.5) 36(54.5) 1.177 0.278 其他 39 22(56.4) 17(43.6) 组织学分级(G) G1~2 51 25(49.0) 26(51.0) 0.010 0.920 G3~4 54 27(50.0) 27(50.0) 肿瘤大小(cm) <5 49 28(57.1) 21(42.9) 2.134 0.144 ≥5 56 24(42.9) 32(57.1) T分期 1~2期 53 35(66.0) 18(34.0) 11.675 0.001 3~4期 52 17(32.7) 35(67.3) N分期 0~1期 60 39(65.0) 21(35.0) 13.414 < 0.001 2~3期 45 13(28.9) 32(71.1) 表 2 Cox回归分析结果
Table 2. The results of the Cox regression analysis
变量 P值 HR(95% CI) CEA 0.021 2.173(1.126~4.192) CA199 0.001 3.144(1.559~6.342) RAB24表达 0.020 2.391(1.148~4.977) T分期 0.039 1.991(1.034~3.832) N分期 0.027 2.106(1.090~4.066) -
[1] SMYTH E C, NILSSON M, GRABSCH H I, et al. Gastric cancer[J]. Lancet, 2020, 396(10251): 635-648. doi: 10.1016/S0140-6736(20)31288-5 [2] HUANG D, SONG M, ABE S K, et al. Family history and gastric cancer incidence and mortality in Asia: a pooled analysis of more than half a million participants[J]. Gastric Cancer, 2024, 27(4): 701-713. doi: 10.1007/s10120-024-01499-1 [3] GYAN W L, HE Y, XU R H. Gastric cancer treatment: recent progress and future perspectives[J]. J Hematol Oncol, 2023, 16(1): 57. DOI: 10.1186/s13045-023-01451-3. [4] PULIGA E, CORSO S, PIETRANTONIO F, et al. Microsatellite instability in gastric cancer: between lights and shadows[J]. Cancer Treat Rev, 2021, 95: 102175. DOI: 10.1016/j.ctrv.2021.102175. [5] DENG J, ZHANG Q, LV L P, et al. Identification of an autophagy-related gene signature for predicting prognosis and immune activity in pancreatic adenocarcinoma[J]. Sci Rep, 2022, 12(1): 7006. DOI: 10.1038/s41598-022-11050-w. [6] YU J F, LANG Q, ZHONG C L, et al. Genome-wide identification of autophagy prognostic signature in pancreatic cancer[J]. Dose Response, 2021, 19(2): 15593258211023260. DOI: 10.1177/15593258211023260. [7] HU D X, JIANG L, LUO S J, et al. Development of an autophagy-related gene expression signature for prognosis prediction in prostate cancer patients[J]. J Transl Med, 2020, 18(1): 160. DOI: 10.1186/s12967-020-02323-x. [8] WU S Y, LIN K C, LAWAL B, et al. MXD3 as an onco-immunological biomarker encompassing the tumor microenvironment, disease staging, prognoses, and therapeutic responses in multiple cancer types[J]. Comput Struct Biotechnol J, 2021, 19: 4970-4983. doi: 10.1016/j.csbj.2021.08.047 [9] HE H, DAI F Y, YU L, et al. Identification and characterization of nine novel human small GTPases showing variable expressions in liver cancer tissues[J]. Gene Expr, 2002, 10(5-6): 231-242. [10] YASUDA T, WANG Y A. Gastric cancer immunosuppressive microenvironment heterogeneity: implications for therapy development[J]. Trends Cancer, 2024, 10(7): 627-642. doi: 10.1016/j.trecan.2024.03.008 [11] 何旭旭, 赵萌, 杨一群, 等. UGT8在胃癌组织中的表达及对胃癌细胞转移的调控作用[J]. 中华全科医学, 2023, 21(4): 544-548. doi: 10.16766/j.cnki.issn.1674-4152.002927HE X X, ZHAO M, YANG Y Q, et al. UGT8 expression in gastric cancer tissues and its regulatory effect on gastric cancer metastasis[J]. Chinese Journal of General Practice, 2023, 21(4): 544-548. doi: 10.16766/j.cnki.issn.1674-4152.002927 [12] WANG Y F, WANG Z W, TANG Y F, et al. Diagnostic model based on key autophagy-related genes in intervertebral disc degeneration[J]. BMC Musculoskelet Disord, 2023, 24(1): 927. DOI: 10.1186/s12891-023-06886-w. [13] JOSHI P, AYYAGARI V, KANDEL S, et al. Loss of RAB25 cooperates with oncogenes in the transformation of human mammary epithelial cells (HMECs) to give rise to claudin-low tumors[J]. Biomed Res Int, 2024, 2024: 8544837. DOI: 10.1155/2024/8544837. [14] SEN P, GANGULY P, KULKARNI K K, et al. Differential transcriptome analysis in HPV-positive and HPV-negative cervical cancer cells through CRISPR knockout of miR-214[J]. J Biosci, 2020, 45: 104. doi: 10.1007/s12038-020-00075-w [15] KUMAR V, RAMNARAYANAN K, SUNDAR R, et al. Single-cell atlas of lineage states, tumor microenvironment, and subtype-specific expression programs in gastric cancer[J]. Cancer Discov, 2022, 12(3): 670-691. doi: 10.1158/2159-8290.CD-21-0683 [16] JIANG Y M, ZHOU K N, SUN Z P, et al. Non-invasive tumor microenvironment evaluation and treatment response prediction in gastric cancer using deep learning radiomics[J]. Cell Rep Med, 2023, 4(8): 101146. DOI: 10.1016/j.xcrm.2023.101146. [17] WEI J, LU X F, LIU Q, et al. Neoadjuvant sintilimab in combination with concurrent chemoradiotherapy for locally advanced gastric or gastroesophageal junction adenocarcinoma: a single-arm phase 2 trial[J]. Nat Commun, 2023, 14(1): 4904. DOI: 10.1038/s41467-023-40480-x. [18] 王星月, 江蕾, 杨俊伟. 巨噬细胞能量代谢与肾脏疾病的研究进展[J]. 中华全科医学, 2020, 18(8): 1348-1352. doi: 10.16766/j.cnki.issn.1674-4152.001504WANG X Y, JIANG L, YANG J W, et al. The role of metabolism in macrophage in kidney diseases[J]. Chinese Journal of General Practice, 2020, 18(8): 1348-1352. doi: 10.16766/j.cnki.issn.1674-4152.001504 [19] KOCH M, GONG R, FRIEDRICH V, et al. CagA-specific Gastric CD8+ tissue-resident T cells control helicobacter pylori during the early infection phase[J]. Gastroenterology, 2023, 164(4): 550-566. doi: 10.1053/j.gastro.2022.12.016 -





 下载:
下载: